A thermostated cell for electrochemistry: minimising natural convection and investigating the role of evaporation and radiation†
Abstract
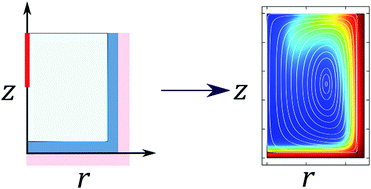
An optimised thermostated electrochemical cell is designed and implemented. This is informed by experimental and computational studies characterizing the extent to which the thermostating of an electrochemical cell via a heated bath can be realised, both with the cell closed and open to the environment. The heat transfer in the system is simulated and probed experimentally; special emphasis is put on heat loss due to radiation and evaporation. Experiments and simulations demonstrate that these two mechanisms of heat transfer lead to a steady temperature in the cell that differs from that of the thermostat by ∼0.1 K. Simulations indicate that spatial inhomogeneities in the stationary temperature drive natural convective flows with a significant velocity. These new physical insights inform the optimization of a new electrochemical cell and its application in measurements of the impact frequency of silver nanoparticles as a function of temperature.


 Please wait while we load your content...
Please wait while we load your content...