Photoreaction of BlrP1: the role of a nonlinear photo-intensity sensor†
Abstract
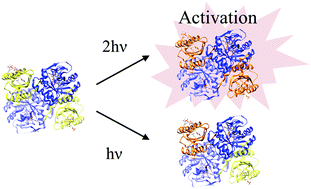
Blue-light-regulated phosphodiesterase 1 (BlrP1) is a blue light sensor protein that controls the hydrolysis of cyclic dimeric guanosine monophosphate, which regulates cellular motility, virulence, and formation of biofilms. In this report, the photoreaction dynamics of BlrP1 and its blue light sensor using a flavin adenine dinucleotide (BLUF) domain were investigated by the time-resolved transient grating method. Only a minor conformational change of the BlrP1–BLUF domain was observed. In contrast, a significant conformational change of the full-length BlrP1 was detected as a diffusion change with a time constant of 21 ms. Interestingly, the extent of the conformational change was concentration-dependent and the dimer form of BlrP1 was found to be the species that exhibited the conformational change. In combination with circular dichroic measurements, a quaternary structural change was determined to be the main origin of the diffusion change. Surprisingly, this conformational change was found to depend strongly on the excitation light intensity. This light-intensity-dependence indicates that the conformational change is induced by the photoexcitation of two monomer units of the dimer. The results suggest that BlrP1 is not only a photosensor but also a light intensity sensor possessing a nonlinear response.


 Please wait while we load your content...
Please wait while we load your content...