Multiscale design of ZnO nanostructured photocatalysts†
Abstract
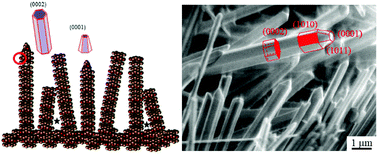
A systematic investigation of the photocatalytic activity (PCA) of nanostructured ZnO films showed how this is directly affected by the films’ morphology at different scales, from the macroscale morphology of films (e.g. thickness and surface area), to the microscale feature arrangement (e.g. aligned vs. randomly oriented structures or interpenetrated ones), to the nanoscale structure (e.g. crystal size and orientation). The interest in immobilizing photocatalysts in water treatment stems from concerns about the potential toxicity of their slurry form, which requires expensive downstream removal. Immobilisation, though, leads to a reduction in PCA, generally attributed to a lower surface area. By reducing the films’ feature size to the nanoscale, an immobilized photocatalyst with high surface area can be achieved. At this scale, however, feature structuring and morphology become important as they determine the interaction between light and the photocatalytic material. In this work, nanostructured ZnO films with different morphology, arrangement and structure were produced by electrochemical anodization of zinc and were tested using the degradation of phenol in a batch reactor as a model system. Results show that the PCA for immobilized catalysts can be optimised by controlling microscale arrangement (light absorbance capacity) and nanoscale structure (crystal size and orientation) rather than macroscale morphology (surface area). These results provide a clear direction to maximising the photocatalytic activity of immobilised photocatalysts for the removal of organic pollutants from water.


 Please wait while we load your content...
Please wait while we load your content...