In situ FTIR studies on the oxidation of isopropyl alcohol over SnO2 as a function of temperature up to 600 °C and a comparison to the analogous plasma-driven process†
Abstract
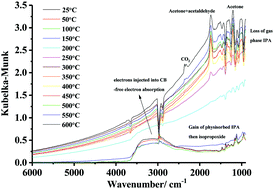
This paper reports the application of in situ reflectance Fourier Transform InfraRed spectroscopy to the study of the thermal and plasma driven reaction of IsoPropyl Alcohol (IPA) at SnO2-coated Macor, the latter a ceramic material comprised of the oxides of Al, Mg and Si. The data so obtained were compared to those obtained using uncoated Macor. When uncoated Macor was employed, no reaction of the IPA was observed up to 600 °C in the thermal experiments, whereas a number of products were observed in the plasma-driven experiments. The results obtained using coated Macor were somewhat different, with no reaction taking place in the plasma-driven experiments, whilst significant reaction took place in the thermally-driven process. In the latter experiments, the chemistry was observed to show four distinct temperature regions, with electron injection into the conduction band of the SnO2 playing a significant role, culminating in the production of CO2. The data were interpreted in terms of a model in which physisorbed IPA was converted to two forms of isopropoxide: this was converted to acetone and acetaldehyde via adsorbed enolate. The data clearly support the catalytic activity of Macor in the plasma-driven conversion of IPA.


 Please wait while we load your content...
Please wait while we load your content...