Ab initio and transition state theory study of the OH + HO2 → H2O + O2(3Σg−)/O2(1Δg) reactions: yield and role of O2(1Δg) in H2O2 decomposition and in combustion of H2†
Abstract
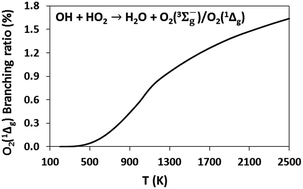
Reactions of hydroxyl (OH) and hydroperoxyl (HO2) are important for governing the reactivity of combustion systems. We performed post-CCSD(T) ab initio calculations at the W3X-L//CCSD = FC/cc-pVTZ level to explore the triplet ground-state and singlet excited-state potential energy surfaces of the OH + HO2 → H2O + O2(3Σg−)/O2(1Δg) reactions. Using microcanonical and multistructural canonical transition state theories, we calculated the rate constant for the triplet and singlet channels over the temperature range 200–2500 K, represented by k(T) = 3.08 × 1012T0.07 exp(1151/RT) + 8.00 × 1012T0.32 exp(−6896/RT) and k(T) = 2.14 × 106T1.65 exp(−2180/RT) in cm3 mol−1 s−1, respectively. The branching ratios show that the yield of singlet excited oxygen is small (<0.5% below 1000 K). To ascertain the importance of singlet oxygen channel, our new kinetic information was implemented into the kinetic model for hydrogen combustion recently updated by Konnov (Combust. Flame, 2015, 162, 3755–3772). The updated kinetic model was used to perform H2O2 thermal decomposition simulations for comparison against shock tube experiments performed by Hong et al. (Proc. Combust. Inst., 2013, 34, 565–571), and to estimate flame speeds and ignition delay times in H2 mixtures. The simulation predicted a larger amount of O2(1Δg) in H2O2 decomposition than that predicted by Konnov's original model. These differences in the O2(1Δg) yield are due to the use of a higher ab initio level and a more sophisticated methodology to compute the rate constant than those used in previous studies, thereby predicting a significantly larger rate constant. No effect was observed on the rate of the H2O2 decomposition and on the flame speeds and ignition delay times of different H2–oxidizer mixtures. However, if the oxidizer is seeded with O3, small differences appear in the flame speed. Given that O2(1Δg) is much more reactive than O2(3Σg−), we do not preclude an effect of the singlet channel of the titled reaction in other combustion systems, especially in systems where excited oxygen plays an important role.


 Please wait while we load your content...
Please wait while we load your content...