Paramagnetic ionic plastic crystals containing the octamethylferrocenium cation: counteranion dependence of phase transitions and crystal structures†
Abstract
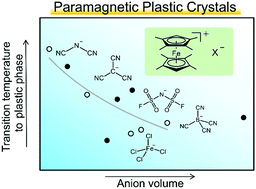
In recent years, ionic plastic crystals have attracted much attention. Many metallocenium salts exhibit plastic phases, but factors affecting their phase transitions are yet to be elucidated. To investigate these factors, we synthesized octamethylferrocenium salts with various counteranions [Fe(C5Me4H)2]X ([1]X; X− = B(CN)4−, C(CN)3−, N(CN)2−, FSA (= (SO2F)2N−), FeCl4−, GaCl4− and CPFSA (= CF2(SO2CF2)2N−)) and elucidated their crystal structures and phase behavior. Correlations between the crystal structures and phase sequences, and the shapes and volumes of the anions are discussed. Except for [1][CPFSA], these salts exhibit phase transitions to plastic phases at or above room temperature (TC = 298–386 K), and the plastic phases exhibit either NaCl- or anti-NiAs-type structures. X-ray crystal structure analyses of these salts at 100 K revealed that they have structures in which cations and anions are alternately arranged, with the exception of [1][CPFSA]. [1][CPFSA] exhibits a structure in which anions and cations are separately stacked to form columns. [1][N(CN)2] exhibits a polar crystal structure that undergoes a monotropic phase transition to a centrosymmetric structure. The magnetic susceptibilities of room-temperature plastic crystals [1][GaCl4] and [1][FeCl4] were investigated; the latter exhibits a small ferromagnetic interaction at low temperatures.
- This article is part of the themed collection: Complex molecular systems: supramolecules, biomolecules and interfaces


 Please wait while we load your content...
Please wait while we load your content...