Comparative studies on conventional and solvent-free synthesis toward hydrazones: application of PXRD and chemometric data analysis in mechanochemical reaction monitoring†
Abstract
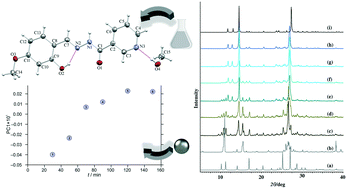
Synthesis of hydrazones was performed via both conventional and solvent-free routes using the corresponding hydrazide (isonicotinic hydrazide, nicotinic hydrazide, 2-aminobenzhydrazide or 4-aminobenzhydrazide) and appropriate aldehyde (salicylaldehyde, 3-methoxysalicylaldehyde or 4-methoxysalicylaldehyde). A systematic study dedicated to solvatomorphism or polymorphism screening resulted in the formation of twelve novel crystalline forms, and eight of these were characterized via the single crystal X-ray diffraction method. In all studied structures, the molecules were assembled into endless supramolecular chains, discrete rings, chains of rings or nets. The mechanochemical synthesis employing liquid-assisted grinding was also applied and the nicotinic- and isonicotinic-based hydrazones were found to form readily from their corresponding precursors. The chemometric study using principal component analysis for mechanochemical synthesis monitoring was implemented for the first time to provide an insight into the reaction profiles. A thoughtful combination of ex situ powder X-ray diffraction and chemometric analysis was essential to identify a stepwise mechanism for the hydrazone formation via an intermediate phase. In five investigated reactions the first principal component accounted for at least 75% of the total variance, whereas in the case of two reactions this component accounted for 69.72 and 46.23% of the total variance. The hydrazones were also evaluated for cytotoxic activity in vitro. All compounds exhibited weak to moderate cytotoxicity against THP-1 and no cytotoxicity against HepG2 cells. Substantial antibacterial activity was obtained against Moraxella catarrhalis while no growth inhibition of Staphylococcus aureus, Enterococcus faecalis and Escherichia coli was observed.
- This article is part of the themed collection: Editor’s Collection: Mechanochemistry


 Please wait while we load your content...
Please wait while we load your content...