Development of a gold nanoparticle enhanced enzyme linked immunosorbent assay based on monoclonal antibodies for the detection of fumonisin B1, B2, and B3 in maize†
Abstract
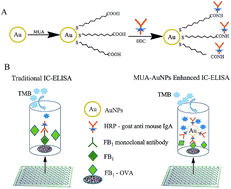
In this paper, three hybridoma cell lines that secrete monoclonal antibodies against fumonisin B1 (FB1), specifically antibody subtypes IgA (heavy-chain) and kappa (light-chain), were obtained by immunization and cell cloning procedures. And the surfaces of gold nanoparticles (AuNPs) were modified with mercaptoundecanoic acid (MUA) to obtain AuNPs–MUA, which was used as a carrier for horseradish peroxidase (HRP)-goat anti-mouse IgA to develop an enhanced indirect competitive enzyme-linked immunosorbent assay (IC-ELISA) for the detection of FB1. The 50% inhibitory concentration (IC50) and the limit of detection (LOD, 15% inhibitory concentration, IC15) of this method were 0.93 ± 0.058 and 0.078 ± 0.013 μg L−1, respectively. The detection sensitivity was approximately 10 times higher than that of the traditional IC-ELISA (IC50 is 9.57 μg L−1). The cross-reactivity rates of AuNP enhanced IC-ELISA with FB1, FB2, and FB3 were 100%, 86.9%, and 79.5%, respectively, and did not cross with other mycotoxins. The total amount of fumonisins (FB1, FB2, and FB3) in actual contaminated maize samples was determined by AuNP enhanced IC-ELISA, and the results were consistent with liquid chromatography-tandem mass spectrometry (LC-MS/MS) with R2 = 0.9989. AuNP enhanced IC-ELISA showed good accuracy and stability, which made it one of the solutions to quantitatively detect the total content of fumonisins (FB1, FB2, and FB3) in maize.


 Please wait while we load your content...
Please wait while we load your content...