Quercetin-coated Fe3O4 nanoparticle sensors based on low-field NMR for determination and removal of Pb2+ and Cu2+ in biological samples†
Abstract
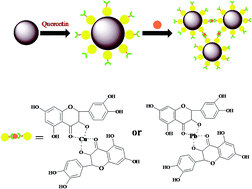
In this paper, we develop quercetin-coated Fe3O4 nanoparticles (QMNPS) as a sensor for the detection and removal of Pb2+ and Cu2+ based on low-field NMR. QMNPS were characterized by TEM, FTIR and XRD. The NMR sensor showed high selectivity toward Pb2+ or Cu2+. The standard curves for ΔT2-ion concentration showed good correlations, i.e., R2 = 0.9912 for Pb2+ and 0.9983 for Cu2+; the linear ranges and the limits of detection were 4.8 × 10−6–1 × 10−4 mol L−1 and 1.6 × 10−6 mol L−1 for Pb2+, and 5.0 × 10−6–1 × 10−4 mol L−1 and 2.0 × 10−6 mol L−1 for Cu2+, respectively. The calculated maximum adsorption capacity was about 71 mg for Cu2+ and 68 mg for Pb2+ per gram of QMMP, which was superior to the results of some previous reports. The detection and removal of Pb2+ or Cu2+ was based on the coordination reaction between the NMR sensor of QMNPs and Pb2+ or Cu2+, which led to the aggregation of QMNPs. As a result, the proposed NMR sensor was successfully applied for the determination and removal of Pb2+ and Cu2+ in contaminated water and urine samples with excellent recoveries and high adsorbent capacities.


 Please wait while we load your content...
Please wait while we load your content...