Ultra-high throughput functional enrichment of large monoamine oxidase (MAO-N) libraries by fluorescence activated cell sorting†
Abstract
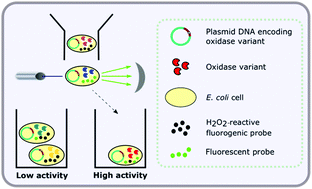
Directed evolution enables the improvement and optimisation of enzymes for particular applications and is a valuable tool for biotechnology and synthetic biology. However, studies are often limited in their scope by the inability to screen very large numbers of variants to identify improved enzymes. One class of enzyme for which a universal, operationally simple ultra-high throughput (>106 variants per day) assay is not available is flavin adenine dinucleotide (FAD) dependent oxidases. The current high throughput assay involves a visual, colourimetric, colony-based screen, however this is not suitable for very large libraries and does not enable quantification of the relative fitness of variants. To address this, we describe an optimised method for the sensitive detection of oxidase activity within single Escherichia coli (E. coli) cells, using the monoamine oxidase from Aspergillus niger, MAO-N, as a model system. In contrast to other methods for the screening of oxidase activity in vivo, this method does not require cell surface expression, emulsion formation or the addition of an extracellular peroxidase. Furthermore, we show that fluorescence activated cell sorting (FACS) of large libraries derived from MAO-N under the assay conditions can enrich the library in functional variants at much higher rates than via the colony-based method. We demonstrate its use for directed evolution by identifying a new mutant of MAO-N with improved activity towards a novel secondary amine substrate. This work demonstrates, for the first time, an ultra-high throughput screening methodology widely applicable for the directed evolution of FAD dependent oxidases in E. coli.


 Please wait while we load your content...
Please wait while we load your content...