Microfluidic single-cell analysis of oxidative stress in Dictyostelium discoideum
Abstract
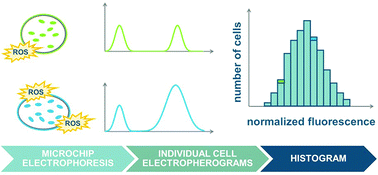
Microfluidic chemical cytometry is a powerful technique for examining chemical contents of individual cells, but applications have focused on cells from multicellular organisms, especially mammals. We demonstrate the first use of microfluidic chemical cytometry to examine a unicellular organism, the social amoeba Dictyostelium discoideum. We used the reactive oxygen species indicator dichlorodihydrofluorescein diacetate to report on oxidative stress and controlled for variations in indicator loading and retention using carboxyfluorescein diacetate as an internal standard. After optimizing indicator concentration, we investigated the effect of peroxide treatment through single-cell measurements of 353 individual cells. The peak area ratio of dichlorofluorescein to carboxyfluorescein increased from 1.69 ± 0.89 for untreated cells to 5.19 ± 2.72 for cells treated with 40 mM hydrogen peroxide. Interestingly, the variance of the data also increased with oxidative stress. While preliminary, these results are consistent with the hypothesis that heterogeneous stress responses in unicellular organisms may be adaptive.


 Please wait while we load your content...
Please wait while we load your content...