In situ protein-templated porous protein–hydroxylapatite nanocomposite microspheres for pH-dependent sustained anticancer drug release†
Abstract
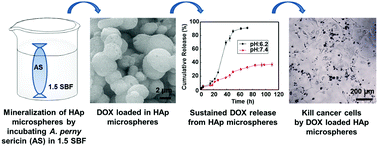
Silk sericin, a water-soluble glue-like protein, is extensively used as a biomaterial because it is biocompatible, hydrophilic, and biodegradable. In addition, hydroxylapatite-based carriers are functionally efficient for drug or gene delivery due to their biodegradability, biocompatibility, and easy metabolism in vivo. Herein, for the first time, this study used sericin, from a wild silkworm called Antheraea pernyi (A. pernyi), as a template to nucleate hydroxylapatite (HAp) nanoneedles and form porous sericin–HAp nanocomposite microspheres as an anticancer drug carrier. Specifically, A. pernyi sericin (AS) was incubated in 1.5× simulated body fluid to induce the formation of porous AS/HAp microspheres in situ. Doxorubicin (DOX) loading and release assays proved that the microspheres exhibited pH-dependent controlled and sustained release of DOX. In particular, the microspheres can selectively release DOX at a higher rate at the acidic conditions typical for tumor microenvironment than at the physiological conditions typical for normal tissues, which will potentially reduce the side effects of the cancer drugs in normal tissues. Cancer cell toxicity assay, cancer cell imaging, and intracellular DOX distribution assay provided further evidence to support the pH-dependent controlled and sustained release of DOX to cancer cells from the microspheres. Our work has demonstrated a biomimetic strategy for the design and synthesis of silk protein-based drug carriers that can be potentially employed in drug delivery and regenerative medicine.


 Please wait while we load your content...
Please wait while we load your content...