Carbon thin-layer-coated manganese oxide nanocrystals as an effective support for high-performance Pt electrocatalysts stabilized at a metal–metal oxide–carbon triple junction†
Abstract
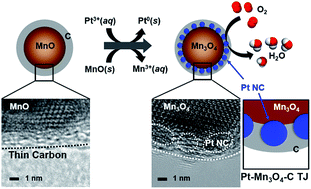
The use of the metal-oxide support has been proved to be an effective approach in the development of fuel-cell catalysts especially for improving the stability of Pt-based electrocatalysts, which is not attainable with conventional carbonaceous materials. Nonetheless, because of the intrinsic low electrical conductivity of pure metal-oxides, metal-oxide-supported systems have not been productive in terms of mass activity that is a primary criterion for the cost-effective electrocatalysts. This paper reports the fabrication of a unique Pt/Mn3O4–carbon triple-junction (TJ) structure, bearing a high density of Pt catalysts entirely sandwiched in between the nano-sized Mn3O4 core and surrounding carbon shell, which integrates the complementary advantages of metal-oxide and carbon supports and thus endows Pt catalysts with optimized electrocatalytic performance. In this study, the unexplored capacity of the Mn3O4 surface was found to accelerate the polymerization process of the surface attached dopamine molecule even under an ordinarily unreactive condition, which enabled the formation of a very thin and contiguous polydopamine layer. The resultant polydopamine film could be converted into a 1.8 nm-thin N-doped carbon layer with sufficiently high electrical conductivity, which allowed the Pt deposition during the galvanic replacement reaction to occur exclusively onto the manganese oxide surface covered underneath the carbon layer and therefore produced the TJ of Pt/Mn3O4–carbon. The resultant TJ structure exhibited the 10-fold enhanced mass activity in the oxygen reduction reaction compared with that of the Mn3O4-supported Pt, which is the highest mass activity for the ORR achieved with oxide-supported catalysts that has been reported so far, while preserving the sufficient durability.


 Please wait while we load your content...
Please wait while we load your content...