In situ growth of cobalt sulfide hollow nanospheres embedded in nitrogen and sulfur co-doped graphene nanoholes as a highly active electrocatalyst for oxygen reduction and evolution†
Abstract
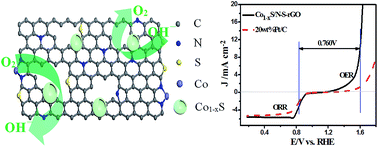
Developing high-performance bifunctional electrocatalysts for the oxygen reduction reaction (ORR) and the oxygen evolution reaction (OER) using nonprecious metal-based catalysts is a major challenge for achieving the commercial success of regenerative fuel cells and rechargeable metal–air batteries. In the present study, we designed a new type of bifunctional catalyst by embedding cobalt sulfide hollow nanospheres in nitrogen and sulfur co-doped graphene nanoholes (Co1−xS/N–S–G) via a simple, one-pot pyrolysis method. The catalyst had a high specific surface area (390.6 m2 g−1) with a hierarchical meso–macroporous structure. In an alkaline medium, the catalyst exhibited high ORR catalytic activity, with a half-wave potential 30 mV more positive and a diffusion-limiting current density 15% higher than a commercial Pt/C catalyst, and the catalyst is also highly active for OER with a small overpotential of 371 mV for 10 mA cm−2 current density. Its overall oxygen electrode activity parameter (ΔE) is 0.760 V, which is smaller than that of Pt/C and most of the non-precious metal catalysts in previous studies. Furthermore, it demonstrated better durability towards both the ORR and OER. Detailed investigation clarified that the material's excellent electrocatalytic performance is attributable to: (1) a synergistic effect, induced by the presence of multiple types of active sites, including cobalt sulfide hollow nanospheres, nitrogen and sulfur dopants, and possible Co–N–C sites; (2) cobalt sulfide hollow nanospheres penetrating through the plane of graphene sheets form strong interaction between them; (3) more edge defects associated with the existence of nanoholes on the graphene basal plane; and (4) the high surface area and efficient mass transfer arising from the hierarchical porous structure.


 Please wait while we load your content...
Please wait while we load your content...