Orthogonal self-assembly of a trigonal triptycene triacid: signaling of exfoliation of porous 2D metal–organic layers by fluorescence and selective CO2 capture by the hydrogen-bonded MOF†
Abstract
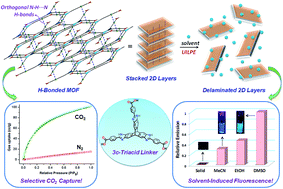
Trigonal 3-connecting imidazole-annulated triptycene triacid (H3TPA) is a molecular module that is programmed for orthogonal self-assembly. Upon treatment with metal salts such as CoCl2, Mn(NO3)2, Zn(NO3)2 and Cd(NO3)2, highly porous isostructural MOFs are obtained in which the TPA linker undergoes metal–ligand coordination and hydrogen bonding through carboxylate groups and imidazole moieties, respectively. The MOFs are constructed by hydrogen bond-mediated offset stacking of porous (3,3) honeycomb layers formed by the self-assembly of TPA with 3-connecting triangular bimetallic SBUs. We show that the interlayer hydrogen bonds can be disrupted by solvents such as DMSO, leading to solvent-induced delamination of 2D metal–organic nanosheets. The delamination is signaled by turn-on of fluorescence, which is suppressed in the bulk state. Indeed, the extent of exfoliation with different solvents – as reflected from fluorescence quantum yields as well as solvent-induced shifts in emission maxima – can be nicely correlated with Gutmann's solvent DN numbers, which are a measure of the ability of solvents to accept hydrogens in hydrogen bonds. The results demonstrate how the bulk materials with layered structures can be (i) engineered in a ‘bottom-up’ approach by orthogonal self-assembly of an organic linker created by de novo design and (ii) in turn be exfoliated in a ‘top-down’ approach by solvent-induced ultrasonication. As bulk materials, the hydrogen-bonded MOFs lend themselves to selective as well as high adsorption of CO2 under ambient conditions as a result of the nitrogenous environment of pores conferred by the benzimidazole moieties.


 Please wait while we load your content...
Please wait while we load your content...