Two in one: N-doped tubular carbon nanostructure as an efficient metal-free dual electrocatalyst for hydrogen evolution and oxygen reduction reactions†
Abstract
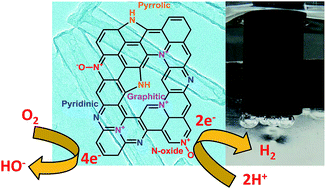
An efficient, N-doped, pore-engineered carbon as a dual electrocatalyst for hydrogen evolution reaction (HER) and oxygen reduction reaction (ORR) is developed with the potential to replace traditional precious metal (Pt)-based catalysts. Rationally designed, N-doped carbons (NDCs) with wider mesopores (15–27 nm) exhibiting superior mass transfer properties were obtained following pyrolysis and demineralization of polydopamine-coated halloysite clay nanotubes. NDCs thus obtained exhibit excellent electrocatalytic activity for hydrogen evolution reaction (HER) with a lower onset potential (117 mV), smaller Tafel slope (94 mV dec−1) and high exchange current density (jo = 1.5 × 10−2 mA cm−2), which is comparable and even superior to other multiple heteroatom-doped carbons and transition metal chalcogenide/oxide-based systems reported previously. Furthermore, NDCs participate in an efficient, direct four-electron pathway for the reduction of molecular oxygen to water. The observed bifunctional electrocatalytic activity of the NDC can be attributed to the synergistic effect of enhanced mass transfer and efficient charge transfer processes at the electrodes and hence deems fit to be a promising candidate for future renewable energy blueprints viz. metal–air batteries and regenerative fuel cell technology.


 Please wait while we load your content...
Please wait while we load your content...