Tunable thermodynamic activity of LaxSr1−xMnyAl1−yO3−δ (0 ≤ x ≤ 1, 0 ≤ y ≤ 1) perovskites for solar thermochemical fuel synthesis†
Abstract
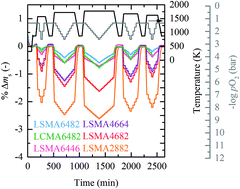
Nonstoichiometric metal oxides with variable valence are attractive redox materials for thermochemical and electrochemical fuel processing. To guide the design of advanced redox materials for solar-driven splitting of CO2 and/or H2O to produce CO and/or H2 (syngas), we investigate the equilibrium thermodynamics of the LaxSr1−xMnyAl1−yO3−δ perovskite family (0 ≤ x ≤ 1, 0 ≤ y ≤ 1) and La0.6Ca0.4Mn0.8Al0.2O3−δ, and compare them to those of CeO2 as the baseline. Oxygen nonstoichiometry measurements from 1573 to 1773 K and from 0.206 to 180 mbar O2 show a tunable reduction extent, increasing with increasing Sr content. Maximal nonstoichiometry of 0.32 is established with La0.2Sr0.8Mn0.8Al0.2O3−δ at 1773 K and 2.37 mbar O2. As a trend, we find that oxygen capacities are most sensitive to the A-cation composition. Partial molar enthalpy, entropy and Gibbs free energy changes for oxide reduction are extracted from the experimental data using defect models for Mn4+/Mn3+ and Mn3+/Mn2+ redox couples. We find that perovskites exhibit typically decreasing enthalpy changes with increasing nonstoichiometries. This desirable characteristic is most pronounced by La0.6Sr0.4Mn0.4Al0.6O3−δ, rendering it attractive for CO2 and H2O splitting. Generally, perovskites show lower enthalpy and entropy changes than ceria, resulting in more favorable reduction but less favorable oxidation equilibria. The energy penalties due to larger temperature swings and excess oxidants are discussed in particular. Using electronic structure theory, we conclude with a practical methodology estimating thermodynamic activity to rationally design perovskites with variable stoichiometry and valence.


 Please wait while we load your content...
Please wait while we load your content...