Site-isolated manganese carbonyl on bipyridine-functionalities of periodic mesoporous organosilicas: efficient CO2 photoreduction and detection of key reaction intermediates†
Abstract
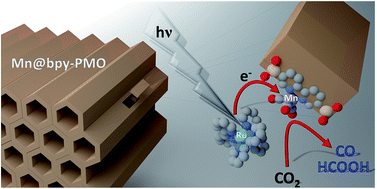
Well-defined and fully characterized supported CO2 reduction catalysts are developed through the immobilization of an earth abundant Mn complex on bpy-PMO (bpy = bipyridine; PMO = Periodic Mesoporous Organosilica) platform materials. The resulting isolated Mn-carbonyl centers coordinated to bipyridine functionalities of bpy-PMO catalyze the photoreduction of CO2 into CO and HCOOH with up to ca. 720 TON in the presence of BIH (1,3-dimethyl-2-phenyl-2,3-dihydro-1H-benzoimidazole), used as the electron donor. A broad range of photochemical conditions (varying solvents, sacrificial electron donors, photosensitizer type and concentration, catalyst loading as well as the Mn loading within the PMO) are investigated, demonstrating high activity even for simple organic dyes and Zn-porphyrin as photosensitizers. Spectroscopic and catalytic data also indicate that site isolation of the Mn complex in the PMO framework probably inhibits bimolecular processes such as dimerisation and disproportionation and thus allows the spectroscopic observation of key reaction intermediates, namely the two meridional isomers of the carbonyl complexes and the bipyridine radical anion species.


 Please wait while we load your content...
Please wait while we load your content...