Selective anomeric acetylation of unprotected sugars in water†
Abstract
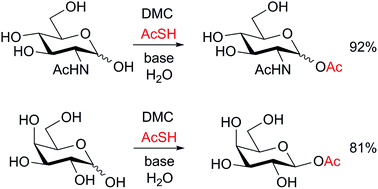
High yielding selective acetylation of only the anomeric hydroxyl of unprotected sugars is possible in aqueous solution using 2-chloro-1,3-dimethylimidazolinium chloride (DMC), thioacetic acid, and a suitable base. The reaction, which may be performed on a multi-gram scale, is stereoselective for sugars that possess a hydroxyl group at position-2, exclusively yielding the 1,2-trans products. The use of an iterative reagent addition procedure allows the use of sodium carbonate as the base, avoiding the formation of triethylammonium salts, which may hamper product purification. The glycosyl acetate products may be used as donor substrates for glycosidase-catalysed synthesis. The crude aqueous acetylation reaction mixture may also be used for this purpose.


 Please wait while we load your content...
Please wait while we load your content...