Structural changes in equimolar ceria–hafnia materials under solar thermochemical looping conditions: cation ordering, formation and stability of the pyrochlore structure†
Abstract
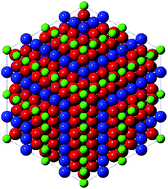
Equimolar ceria–hafnia oxides form a pyrochlore structure Ce2Hf2O7, which exhibits an ordered arrangement of Ce3+ and Hf4+ cations under the reducing conditions of a solar thermochemical looping reactor for the two-step dissociation of water or carbon dioxide. The ceria–hafnia pyrochlore phase was prepared from oxidized ceria–hafnia powders by chemical reduction in a flow of H2/He and by auto-reduction in a flow of Ar at up to 1825 K. Full conversion of Ce4+ to Ce3+ was confirmed by thermogravimetric analysis and Ce K edge X-ray absorption spectroscopy. X-ray diffraction and Hf K edge X-ray absorption spectroscopy identified the pyrochlore phase. The dynamics of the structural changes were determined by time-resolved in situ Ce K edge X-ray absorption spectroscopy and in situ X-ray diffraction. Under the oxidizing conditions of the regeneration step of isothermal carbon dioxide splitting at 1800 K, the pyrochlore transformed to a mixture of fluorite-type or tetragonal ceria and monoclinic and orthorhombic hafnia phases. A κ-Ce2Hf2O8 phase, an oxidized form of the pyrochlore with an ordered arrangement of cations, was not detected.


 Please wait while we load your content...
Please wait while we load your content...