Synthesis of poly(p-phenylene) containing a rhodamine 6G derivative for the detection of Fe(iii) in organic and aqueous media†
Abstract
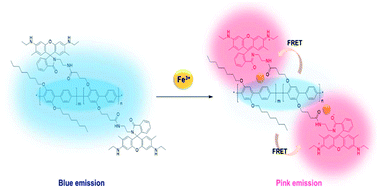
A conjugated polymer (CP) of poly(p-phenylene) (PPP) containing rhodamine 6G (R6G) was synthesized by the Suzuki-coupling reaction, in which PPP acted as a blue-emitting energy donor and R6G acted as a ligand for Fe(III) as well as the energy acceptor for Förster resonance energy transfer (FRET). The spirolactam ring in R6G was nonfluorescent and colorless, but became open upon exposure to Fe(III) and readily absorbed the blue emission of the PPP backbone. As the polymer was exposed to Fe(III), the blue emission from PPP decreased and the red emission of the ring-opened R6G increased via FRET. The sensing of the polymer for Fe(III) in ethanolic solution was not successful, because of negligible FRET. In the solid state of the polymer, the distance between the main chain (PPP) and the side chain (R6G) was sufficiently decreased to enable the transfer of fluorescence energy. As a result, the film sensor could detect Fe(III) in an ethanol solution and a paper-based strip sensor could detect Fe(III) in an aqueous solution with a change in the fluorescence color. The cellulosic paper of the strip provided rapid detection and improved the limit of detection. From the changes in the fluorescence of the polymer sensor in the presence of Fe(III), the film and strip sensors could detect Fe(III) in any media. Moreover, the film could be regenerated for repeated use after treatment with ethylenediaminetetraacetic acid (EDTA).


 Please wait while we load your content...
Please wait while we load your content...