Steric effect on Li+ coordination and transport properties in polyoxetane-based polymer electrolytes bearing nitrile groups†
Abstract
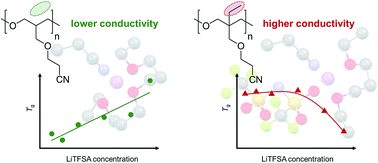
Polymer electrolytes (PEs) that consist of polyoxetane with nitrile side chains and lithium bis(trifluoromethanesulfonyl)amide were studied as electrolytes for Li secondary batteries. Our previous study showed that Li+ ion coordination with the polar nitrile groups tethered to the main chain plays a crucial role in enhancing the Li+ ion transport properties in poly(3-(2-cyanoethoxymethyl)-3-ethyloxetane) (PCEO). Herein, we report the effects of the absence of ethyl groups on the main chain of PCEO on Li+ ion coordination, thermal properties, and ionic transport, by employing a structural analog, poly(3-(2-cyanoethoxymethyl)-oxetane). We found that the steric hindrance derived from the ethyl groups in PCEO resulted in more-pronounced Li+ coordination with the nitrile side chains, and thereby more effective Li+ ion transport was achieved. We also demonstrated successful charge/discharge cycling of a Li/LiFePO4 cell using nitrile-functionalized PE.


 Please wait while we load your content...
Please wait while we load your content...