Synthesis, characterization, and properties of nickel–cobalt layered double hydroxide nanostructures
Abstract
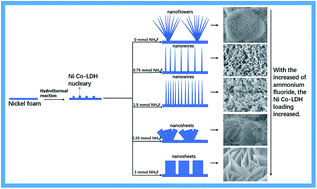
Nickel–cobalt layered double hydroxides (Ni–Co-LDH) have recently been examined for their potential as battery-type hybrid supercapacitors made from metal hydroxide electrode materials, due to their unique spatial structure, excellent electrochemical activity, and good electrical conductivity. However, the main disadvantage restricting the application of Ni–Co-LDHs is their low electronic conductivity, which results in low capacitance. To address this problem, we used different concentrations of ammonium fluoride to control the Ni–Co-LDH surface morphology and direct growth on Ni foam (NF). We created Ni–Co-LDH composite electrode materials with different morphologies that showed large surface areas, high conductivity, and high electrochemical performance. Results showed that the samples prepared with ammonium fluoride additive had a higher specific capacity of about 1445 F g−1 at a current density of 2 A g−1, a good specific capacitance rate of about 59.5% from 2 A g−1 to 40 A g−1, and good capacity retention of up to 99% when the current density was enhanced to 30 A g−1, suggesting promise for future applications.


 Please wait while we load your content...
Please wait while we load your content...