Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates†
Abstract
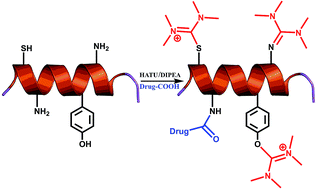
Peptide coupling reagents and especially uronium/guanidinium salts have been extensively utilized in solid-phase peptide synthesis. However, the impact of these reagents in solution phase synthesis, normally used in the formation of peptide-drug conjugates (PDCs), has not been fully explored. Herein, we identified that when guanidinium salts are used in classical peptide coupling conditions, besides leading to the formation of amide bonds, a uronium derivative can also be installed on specific amino acid scaffolds. The formation of this side product depends on the reaction conditions, as also on the nucleophilicity of the susceptible groups. Conditions to avoid this side product formation and a putative reaction mechanism describing its formation are reported.
- This article is part of the themed collection: Editors' collection: Physical Chemistry of Colloids and Interfaces


 Please wait while we load your content...
Please wait while we load your content...