Remarkable dielectric properties of 1 : 2 inter-molecular compound of 2-(4-(dimethylamino) benzylideneamino) benzoic acid and urea due to excited-state intramolecular proton transfer†
Abstract
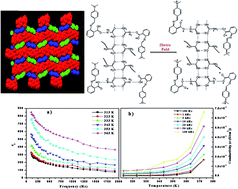
The phase diagram of 2-(4-(dimethylamino) benzylideneamino) benzoic acid (DMABAB)–urea (U) gives two eutectics E1 (mp 107.0 °C) and E2 (mp 138.0 °C) with 0.01 and 0.86 mole fractions of urea, respectively, and a 1 : 2 inter-molecular compound (IMC) with a congruent melting point of 219.0 °C, which has a high dielectric constant (ε = 0.9 × 103) and reasonable electrical conductivity in the order ∼5 × 10−6 S m−1. This is due to the remarkable packing of U molecules in the IMC, where it shows coupling of two different types of cyclic hydrogen-bonded motifs alternating to form a corrugated sheet or chain of rings extending along the b axis via N–H⋯O hydrogen bonding interactions. In the presence of an electric field, an inter-molecular proton transfer process occurs in the NH⋯O bonds of the chain of the U molecules, which leads to disproportionate defects and the formation of polar domains on the macroscopic scale. An appropriate quantity of the IMC is synthesized via a green synthetic method, which has a monoclinic crystal system with the P21/c centrosymmetric space group. Additionally, its crystal data and hydrogen bonding parameters are determined. According to powder X-ray diffraction, spectral and thermal characterization, the eutectics are a mechanical mixture of the IMC and pure components, although the IMC behaves as a pure compound. The IMC shows a broad emission band in the range of 350 to 580 nm with a quantum yield of almost 1 (0.99) upon excitation at the λmax absorption (300 nm) in MeOH solution at a concentration of 1 × 10−5 M.


 Please wait while we load your content...
Please wait while we load your content...