Role of hydrophilic groups in acid intercalated graphene oxide as a superionic conductor
Abstract
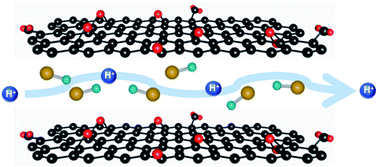
Hybrid materials that are obtained from intercalation of different polar and hydrophilic acid guest molecules within graphene oxide (GO) have been found to exhibit high proton conductivity. The accommodation of these guest precursors within the GO interlayer takes place through weak physical forces. PXRD patterns with enhanced interlayer distance confirmed successful intercalation processes. Raman and IR spectral data reveal the absence of any covalent bond between the functional groups of the guest and the GO host. TGA data confirms improved water adsorbing capacity of the hybrids. At ambient conditions and 90% relative humidity (RH), a RH dependent impedance study shows that intercalation of formic acid (FA) and phosphoric acid (PA) within GO results in ∼7 times higher proton conductivity compared with that for the pristine GO sample. The low activation energy values suggest that proton conduction in the samples is aided by the Grotthuss mechanism. Improvement in the water adsorbing capacity is primarily responsible for such high proton conductivity. The current study suggests that cheap and environmentally friendly GO-based intercalated hybrid materials, such as GO/PA and GO/FA with enhanced proton conductivity, can be appropriate replacements for the expensive Nafion-based solid electrolytes.


 Please wait while we load your content...
Please wait while we load your content...