Cu nanoparticles immobilized on montmorillonite by biquaternary ammonium salts: a highly active and stable heterogeneous catalyst for cascade sequence to indole-2-carboxylic esters†
Abstract
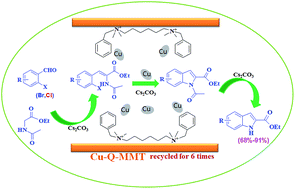
Copper nanoparticles immobilized on montmorillonite (MMT) by biquaternary ammonium salts (N1,N6-dibenzyl-N1,N1,N6,N6-tetramethylheptane-1,6-diaminium bromide, Q) were prepared by cation-exchange and impregnation–reduction and designated Cu-Q-MMT. The material was extensively characterized by various characterization techniques such as FTIR, XRD, XPS, SEM, TEM, and N2 adsorption–desorption. The Cu-Q-MMT could be used as a highly active heterogeneous catalyst for cascade sequence to indole-2-carboxylic esters from ortho-bromobenzaldehydes with ethyl acetamidoacetate. Even for inactive chlorobenzaldehydes, a good yield could be obtained. In addition, the catalyst can be reused six times without any significant loss of activity. The high activity and stability of the Cu-Q-MMT catalyst is mainly attributed to the excellent synergistic effects of biquaternary ammonium salts, Cu nanoparticles and the nanospace structure of MMT.


 Please wait while we load your content...
Please wait while we load your content...