Inhibition effect of aromatic aldehydes on butanol fermentation by Clostridium acetobutylicum
Abstract
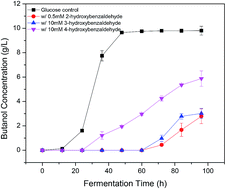
A large number of degradation compounds are formed during biomass pretreatment and they significantly inhibit the efficiency of biomass conversion to biofuels. Of those identified potential inhibitors, aromatic aldehydes play an important role in inhibition activity. Hence the effect of 13 aromatic aldehydes on acetone–butanol–ethanol (ABE) fermentation was assessed at four concentrations in the present work. It was found that the inhibition severity was affected by the ortho substituents (OH > OCH3 > CHO) and strongly related to the position of hydroxyl group instead of the number of hydroxyl groups. The ortho-hydroxyl group significantly contributed to the aromatic aldehyde inhibition. The ortho-substituted 2-hydroxybenzaldehyde caused at least 20-fold stronger inhibition than meta- and para-substituted analogues of 3- and 4-hydroxybenzaldehydes. The presence of ortho-hydroxyl group can form an intramolecular hydrogen bond with carbonyl hydrogen and potentially increase the cell membrane permeability and electrophilicity. Quantitative structure–activity relationship (QSAR) analysis was used to establish a correlation between inhibition activity (IC50) and physicochemical descriptors. A strong correlation was observed between IC50 and the energy of the highest occupied molecular orbital EHOMO.


 Please wait while we load your content...
Please wait while we load your content...