Evaluation of (E)-1,2-di(furan-2-yl)ethene as building unit in diketopyrrolopyrrole alternating copolymers for transistors†
Abstract
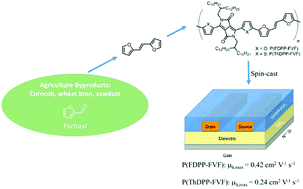
Alternating copolymers, poly(2,5-bis(2-decyltetradecyl)-3,6-di(furan-2-yl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione-b-(E)-1,2-di(furan-2-yl)ethene) (P(FDPP-FVF)) and poly(2,5-bis(2-decyltetradecyl)-3,6-di(thiophen-2-yl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione-b-(E)-1,2-di(furan-2-yl)ethene) (P(ThDPP-FVF)), composed of (E)-1,2-di(furan-2-yl)ethene and diketopyrrolopyrrole, were synthesized and studied for solution-processable organic field effect transistors (OFETs). Highest hole mobilities of 0.45 and 0.24 cm2 V−1 s−1 were measured for P(FDPP-FVF) and P(ThDPP-FVF), respectively, in bottom gate-bottom contact transistor devices. Atomic force microscopy and grazing incidence X-ray diffraction experiments were employed to investigate the crystallinity and morphology for the spin-cast polymer films. It is noteworthy that the thermally annealed P(FDPP-FVF) film exhibited higher crystallinity and small π-stacking distance of 3.47 Å. Moreover, considering the easy synthesis from renewable furfural and better performance of P(FDPP-FVF), our results provide an alternative design strategy for the synthesis of solution-based semiconducting polymers from renewable furan derivatives for OFETs.


 Please wait while we load your content...
Please wait while we load your content...