Colorless polyimides derived from 2R,5R,7S,10S-naphthanetetracarboxylic dianhydride†
Abstract
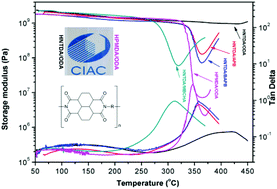
A novel alicyclic dianhydride, 2R,5R,7S,10S-naphthanetetracarboxylic dianhydride (HNTDA), was prepared through the hydrogenation of tetramethyl 1,4,5,8-naphthalenetetracarboxylate, followed by deprotection and dehydration reactions. Single crystal X-ray diffraction results revealed that HNTDA possesses an irregular L-shaped architecture with one anhydride ring equatorial, and the other axial on the naphthane ring. Conventional one-step solution polycondensation of HNTDA with commercial diamines enabled the preparation of fully or semi-alicyclic polyimides with sufficient molecular weights. The properties of HNTDA-based polyimides were systematically characterized and compared with their counterparts based on 1S,2R,4S,5R-cyclohexanetetracarboxylic dianhydride (HPMDA). Dynamic mechanical thermal analysis, thermogravimetric analysis, thermal mechanical analysis, and tensile testing indicated that HNTDA-based polymers exhibit markedly higher thermal and mechanical properties compared with HPMDA-based ones. These improvements can be rationalized according to the rigid, bulky, fused naphthanetetracarboxydiimide moiety, and the pronounced inter- and intra-molecular interactions. Furthermore, HNTDA-based polyimides were colorless or pale-yellow, and displayed good optical transparency, although their cutoff wavelength values were higher than those of HPMDA-based ones.


 Please wait while we load your content...
Please wait while we load your content...