Iridium-catalyzed polymerization of benzoic acids and internal diynes: a new route for constructing high molecular weight polynaphthalenes without the constraint of monomer stoichiometry†
Abstract
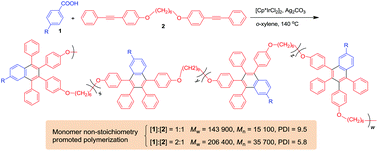
The development of new polymerization reactions conducted under monomer non-stoichiometric conditions has great academic and industrial significance. Herein we report such a new route for synthesizing functional polynaphthalenes (PNs). The polycoupling reactions of benzoic acid and its derivatives (1) with 4,4′-(1,6-hexylenedioxy)bis(diphenylacetylene) (2) proceed smoothly in o-xylene in the presence of [Cp*IrCl2]2 and Ag2CO3 at 140 °C, generating multisubstituted PNs with high molecular weights (Mw up to 228 700) in high yields (up to 98%). The effect of monomer stoichiometry on the polymerization was systematically investigated and the best result was obtained at a molar feed ratio of 1 : 2 of 2 : 1. The associated mechanism for the monomer non-stoichiometry enhancement effect was proposed. All the obtained PNs show good thermal stability and excellent optical transparency. They possess good film-forming ability and their thin films exhibit high refractive indices (n = 1.7653–1.6354). Polymers constructed from non-emissive monomers show light emission in solutions due to the generation of fluorophores in situ during the polymerization. The polymer carrying a tetraphenylethene motif is weakly emissive in solution but emits intensely when aggregated, demonstrating a phenomenon of aggregation-induced emission. The efficient solid-state emission and good photosensitivity enable it to be utilized as a material for fabricating two-dimensional fluorescent photopatterns by irradiating its thin films under UV light through copper masks.


 Please wait while we load your content...
Please wait while we load your content...