Synthesis of 2-amino- and 2-arylazoazulenes via nucleophilic aromatic substitution of 2-chloroazulenes with amines and arylhydrazines†
Abstract
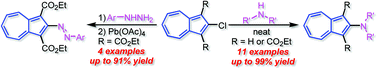
The SNAr reaction of 2-chloroazulene derivative 1 with ethoxycarbonyl groups at the 1,3-positions of the azulene ring with several amines afforded the corresponding 2-aminoazulenes 3–9 in excellent yields. 2-Chloroazulene (2) without the electron-withdrawing groups reacted with highly nucleophilic cyclic amines (i.e., morpholine, piperidine and pyrrolidine) under the high-temperature conditions in a sealed tube to produce the corresponding 2-aminoazulenes 10–12 in good yields. 2-Aminoazulenes 10–14 without the electron-withdrawing groups were also obtained in good yields by the treatment of compounds 3–7 with 100% H3PO4, but in the cases of the reaction of 8 and 9 with a secondary amine function, the decomposition of the products resulted. The synthesis of 2-arylazoazulenes 15–18 was also established via the SNAr reaction of 1 with arylhydrazines. The optical and electrochemical properties of the 2-arylazoazulene derivatives were examined by UV/Vis spectroscopy, theoretical calculations and voltammetric experiments.


 Please wait while we load your content...
Please wait while we load your content...