Superior acidic catalytic activity and stability of Fe-doped HTaWO6 nanotubes†
Abstract
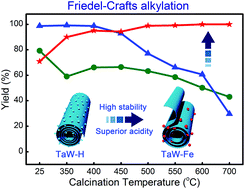
Fe-doped HTaWO6 (H1−3xFexTaWO6, x = 0.23) nanotubes as highly active solid acid catalysts were prepared via an exfoliation–scrolling–exchange process. The specific surface area and pore volume of undoped nanotubes (20.8 m2 g−1, 0.057 cm3 g−1) were remarkably enhanced through Fe3+ ion-exchange (>100 m2 g−1, 0.547 cm3 g−1). Doping Fe ions into the nanotubes endowed them with improved thermal stability due to the stronger interaction between the intercalated Fe3+ ions and the host layers. This interaction also facilitated the preservation of effective Brønsted acid sites and the generation of new acid sites. The integration of these functional roles resulted in Fe-doped nanotubes with high acidic catalytic activities in the Friedel–Crafts alkylation of anisole and the esterification of acetic acid. Facile accessibility to active sites, generation of effective Brønsted acid sites, high stability of the tubular structure and strong acid sites were found to synergistically contribute to the excellent acidic catalytic efficiency. Additionally, the activity of cycled nanocatalysts can be easily recovered through annealing treatment.


 Please wait while we load your content...
Please wait while we load your content...