A pH-responsive sequential-disassembly nanohybrid for mitochondrial targeting†
Abstract
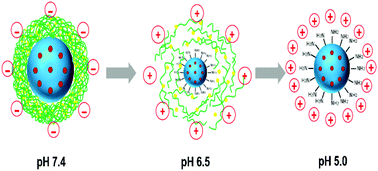
Cationic materials have been reported as promising tools for targeting to mitochondria which are the “power houses” and “metabolic garbage keepers” of cells. However, their positive nature also restricts their in vivo application due to the quick clearance. Herein, we fabricated a nanohybrid consisting of the pH-responsive N-(2-hydroxypropyl)methacrylamide (HPMA) co-polymer (R-P) shells and positive mesoporous silica nanoparticle cores via electrostatic interaction. The anticancer drug, docetaxel (DTX), was encapsulated in the positive MSN cores (MSN–DTX). Once concealed by the anionic R-P shield, the assembled nanohybrid R-P@MSN–DTX will achieve prolonged blood circulation thereby leading to an enhanced EPR effect. At mildly acidic tumor environmental pH, first-stage charge reversion took place due to the hydrolysis of the amide bond on HPMA co-polymers. The de-attachment of the HPMA co-polymer occurred because of the positive charge repulsion and partial exposure of the positively charged MSN core promoted the cell internalization. The second-stage pH-responsiveness in the endo/lysosomes with a more acidic environment accelerates the disassembly of the nanohybrid and the leakage of the core facilitated the endo/lysosome escape and mitochondrial targeting with the help of intracellular compartmental acidity. Gathering up the characteristics of neutralized charge and stepwise pH-responsiveness, the R-P@MSN–DTX acquired a good tumor inhibition rate of 72.6% on nude mice. Our report provided a reference for systemic mitochondrial targeting achieved by the union of “assembly–disassembly”.

 Please wait while we load your content...
Please wait while we load your content...