Covalent modification of biological targets with natural products through Paal–Knorr pyrrole formation
Abstract
Covering: up to June 2017
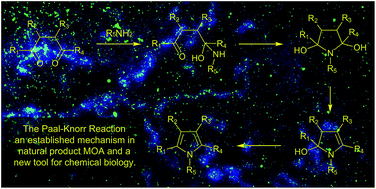
Natural products and endogenous metabolites engage specific targets within tissues and cells through complex mechanisms. This review examines the extent to which natural systems have adopted the Paal–Knorr reaction to engage nucleophilic amine groups within biological targets. Current understanding of this mode of reactivity is limited by only a few examples of this reaction in a biological context. This highlight is intended to stimulate the scientific community to identify potential research directions and applications of the Paal–Knorr reaction in native and engineered biological systems.


 Please wait while we load your content...
Please wait while we load your content...