Improving the photocatalytic activity of polyaniline and a porphyrin via oxidation to obtain a salt and a charge-transfer complex†
Abstract
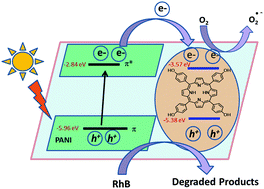
Environmental problems caused by organic dyes have led to the development of efficient methods for their treatment. Photocatalytic degradation has proved to be a viable technique to decompose these pollutants into less dangerous compounds. In this study, the photocatalytic activity of a polyaniline salt and a porphyrin was improved by preparing a hybrid of both components, i.e., a polyaniline-sulfate-5,10,15,20-tetrakis(4-hydroxy phenyl)-21H,23H porphine salt (PANI–H2SO4–TKHP). This salt was synthesized by a chemical oxidative polymerization pathway and used as a photocatalyst for the degradation of RhB dye. The formation of this salt was confirmed by FT-IR, XRD and UV-vis spectroscopy. Oxidation of aniline in the presence of TKHP by ammonium persulfate results in the formation of both the polyaniline salt (PANI–H2SO4–TKHP) and a charge-transfer complex between PANI and TKHP via π–π stacking. PANI–H2SO4–TKHP was observed in a nanotube morphology (which was confirmed by SEM and TEM) with a reasonably good yield (88 wt%), conductivity (1.2 ohm−1 cm−1) and thermal stability (200 °C). The photocatalytic performance of PANI–H2SO4–TKHP was compared with its individual components of PANI–H2SO4 and TKHP under visible light irradiation. PANI–H2SO4–TKHP exhibited an effective photocatalytic activity (78% dye degradation), and a plausible mechanism for photocatalytic degradation was proposed in terms of the HOMO and LUMO levels of the components.


 Please wait while we load your content...
Please wait while we load your content...