Synergistic effect of gold supported on redox active cerium oxide nanoparticles for the catalytic hydrogenation of 4-nitrophenol†
Abstract
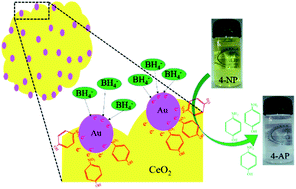
Recently, the conversion of 4-nitrophenol to 4-aminophenol via metal-supported oxide catalysts has attracted significant attention because of its potential applications. The main objective of this study was to demonstrate the crucial role of support materials in heterogeneous catalysts with inherent redox cycling features. The metals supported on these redox active materials can significantly alter the catalytic performance in a synergistic manner. For this, the catalytic hydrogenation of Au–ceria, prepared by altering the annealing–impregnation procedures, was investigated in terms of the size of gold and support material and their redox chemistry. The study clearly shows the variation in the catalytic activity of gold supported on the as-prepared and annealed ceria surface. This variation was due to the fact that the mutual interactions at the interface between absorbed Au and cerium oxide were certainly different in the as-prepared and annealed ceria surfaces with respect to surface chemistry and size of the particles. The redox nature of ceria and partial oxidation of gold on the surface of CeO2 were involved in the catalytic reduction process.


 Please wait while we load your content...
Please wait while we load your content...