Series of high spin mononuclear iron(iii) complexes with Schiff base ligands derived from 2-hydroxybenzophenones†
Abstract
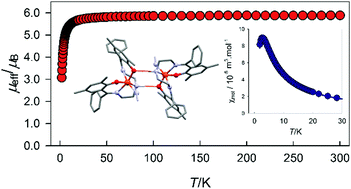
The reaction of various phenols with benzoyl chloride afforded the derivatives of phenyl benzoate that subsequently underwent Fries rearrangement. The obtained 2-hydroxybenzophenone analogues were combined with linear aliphatic triamines, which afforded pentadentate Schiff base ligands. Moreover, nine new iron(III) complexes with the general formula [Fe(Ln)X] (where, Ln is the dianion of the pentadentate Schiff base ligand, N,N′-bis((2-hydroxy-5-methylphenyl)phenyl)methylidene-1,5-diamino-3-azapentane = H2L1, N,N′-bis((2-hydroxy-3,5-dimethylphenyl)phenyl)methylidene-1,5-diamino-3-azapentane = H2L2, N,N′-bis((2-hydroxy-5-chlorophenyl)phenyl)methylidene-1,5-diamino-3-azapentane = H2L3, N,N′-bis((2-hydroxy-4-methylphenyl)phenyl)methylidene-1,5-diamino-3-azapentane = H2L4, N,N′-bis((2-hydroxy-5-bromophenyl)phenyl)methylidene-1,7-diamino-4-azaheptane = H2L5, N,N′-bis((2-hydroxy-5-bromophenyl)phenyl)methylidene-1,7-diamino-4-methyl-4-azaheptane = H2L6 and X is the chlorido, azido or isocyanato terminal ligand) were synthesized and characterized via elemental analysis, and IR and UV-VIS spectroscopy; in addition, the crystal structures of all the complexes were determined by X-ray diffraction. Magnetic investigation reveals high spin state behaviour in all the reported compounds. DFT calculations and analysis of the magnetic functions allowed to extract absolute values of the zero field splitting parameters and exchange coupling constants.


 Please wait while we load your content...
Please wait while we load your content...