Novel organotin(iv) complexes derived from 4-fluorophenyl-selenoacetic acid: synthesis, characterization and in vitro cytostatic activity evaluation†
Abstract
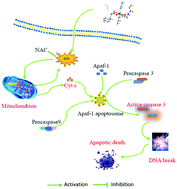
A cluster of novel organotin(IV) complexes with the formulas [Me3Sn(O2CCH2SeC6H4F-p)]n (1), [(Me2Sn)4(μ3-O)2(O2CCH2SeC6H4F-p)4] (2), [n-Bu3Sn(O2CCH2SeC6H4F-p)]n (3) and [n-Bu2Sn(O2CCH2SeC6H4F-p)2] (4) were designed, synthesized, and characterized by elemental analysis, FT-IR, and NMR (1H, 13C, and 119Sn) spectroscopy as well as single-crystal X-ray diffraction. Complexes 1 and 3 represent one-dimensional (1D) coordination polymers composed of alternate [Sn–O]2. The molecular spatial structure of 2 revealed it to be tetranuclear possessing a ladder-like structure and containing four carboxylate ligands. The 1D chain was formed by C–H⋯F interaction. Complex 4 was a monomer connected into a 1D coordination polymer through the intermolecular interaction C–H⋯O. Meanwhile, four novel corresponding organosulfurtin complexes (5–8) were also prepared. Furthermore, complexes 1–4 and 5–8 were evaluated for their in vitro cytostatic activity against human breast cancer cell lines (MDA-MB-231). The preliminary screening showed that the organotin derivatives with an n-butyl group (3, 4 and 7, 8) exhibited significantly higher cytostatic activity than those with a methyl group (1, 2 and 5, 6). Mechanically, further cytotoxic assessments of complex 4 in mediated MDA-MB-231 cells were conducted, and the results revealed that the anti-proliferative and pro-apoptotic effect of complex 4 in MDA-MB-231 cells prominently contributed to the boosted ROS, collapsed mitochondrial membrane potential and the release of cytochrome c from the outer membrane of the mitochondria, which sequently activated caspase-3, the executioner, which was responsible for complex 4's chemotherapeutic potential against MDA-MB-231 cells.


 Please wait while we load your content...
Please wait while we load your content...