Enhanced removal of cephalosporin based antibiotics (CBA) from water by one-pot electrosynthesized Mg(OH)2: a combined theoretical and experimental study to pilot scale
Abstract
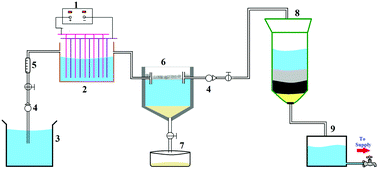
This study explores the removal of cephalosporin based antibiotics (CBA), such as cefepime, cefaclor, cefuroxime, cefazolin, cefixime, cefalexin and ceftobiprole, from water using Mg(OH)2 synthesised using a facile one-pot process via an electrodissolution method. This process is a novel process, where the adsorbents are generated in situ, which is efficient and cost-effective for removing organic pollutants. The adsorption performance for CBA onto Mg(OH)2 was systematically investigated and the experimental results indicated that Mg(OH)2 showed excellent adsorption capacity towards CBA due to the high specific surface area (60.703 m2 g−1) of Mg(OH)2. To optimise the removal efficiency, the effects of contact time, concentration, pH and current density were studied. The adsorption kinetics were modelled using pseudo first and second order kinetics, and Elovich, and Weber and Morris intraparticle diffusion models. The rate constants for all these kinetic models were calculated and the results show that the second order kinetic models were best fitted to model the kinetic adsorption of CBA. Langmuir, Freundlich, D–R isotherm and Temkin models were applied to describe the equilibrium isotherm models and the isotherm constants were determined. The adsorption was studied thermodynamically, and the Gibbs free energy change (ΔG°), enthalpy change (ΔH°), and entropy change (ΔS°) were calculated. The pilot scale study shows that the process is technologically feasible. The adsorbed Mg(OH)2 was characterized using different techniques, such as FESEM, EDAX, XRD, BET and FTIR measurements.


 Please wait while we load your content...
Please wait while we load your content...