Hydrogenation of dimethyl malonate to 1,3-propanediol catalyzed by a Cu/SiO2 catalyst: the reaction network and the effect of Cu+/Cu0 on selectivity†
Abstract
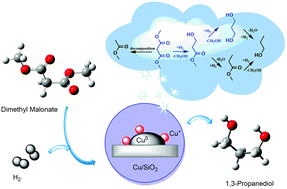
1,3-Propanediol, a vital monomer for the manufacture of commodity polytrimethylene-terephthalate, is commercially produced nowadays through either hydration of acrolein or hydroformylation of ethylene oxide. Herein, for the first time, we present the investigation of an alternative route for 1,3-propanediol production from vapor-phase catalytic hydrogenation of syngas-derived dimethyl malonate on a Cu/SiO2 catalyst. The catalytic reaction network has been disclosed for the Cu/SiO2 catalyst, and the reaction proceeds through sequential hydrogenation with methyl 3-hydroxypropionate as the primary product, which can be further converted into 1,3-propanediol or methyl propionate. Excessive hydrogenation of 1,3-propanediol or methyl propionate leads to the formation of n-propanol. Meanwhile, a small amount of dimethyl malonate cracks into methyl acetate. The structural and textural properties of Cu/SiO2 catalysts with varied copper loadings were extensively characterized by X-ray diffraction, Fourier transform infrared spectroscopy, H2-temperature programmed reduction, X-ray photoelectron spectroscopy, N2 physisorption, CO chemisorption, N2O titration, and transmission electron microscopy. A correlation of the areal activity to copper components suggests that metallic copper modified by the co-present Cu+ species may be the active site for the hydrogenation. The highest 1,3-propanediol selectivity was achieved on a catalyst with a maximum Cu+/(Cu0 + Cu+) ratio of 0.41. The revelation of catalytic networks and insights into the active species can provide guidance for future rational design of catalysts for regioselective hydrogenation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds in dimethyl malonate.
O bonds in dimethyl malonate.


 Please wait while we load your content...
Please wait while we load your content...