Wavelength-focusing organic molecular materials with diazoacetate or fumarate as a monofluorophore†
Abstract
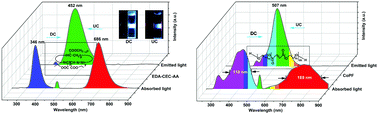
The wavelength of incident light can be shifted by down-conversion (DC) and up-conversion (UC) photofluorescence. DC and UC molecules are typically independent one-way conversion species, and UC should be stimulated by a laser source. In this paper, we find that diazoacetate- and fumarate-containing fluorescent molecules are wavelength-focusing species of simulated solar radiation. Monochromatic UV and NIR lights (354/696, 457/819 or 346/686 nm) are synchronously focused to 454, 473 or 454 nm, respectively, using (5R)-5-cyano-3-carbmenthoxy-2-pyrazoline, 3-carbethoxy-2-pyrazoline grafted polyvinyl alcohol and a cyclic oligomer of ethyl diazoacetate with carbethoxy carbene and allyl acrylate. With a broadband light source, portions of the 300–500 nm and 580–800 nm wavelength bands of incident light are synchronously converted to 507 nm using a copolymer of PEG and fumarate, which offers a promising way to harvest the UV and NIR portions of sunlight in the visible region.


 Please wait while we load your content...
Please wait while we load your content...