A liver microphysiological system of tumor cell dormancy and inflammatory responsiveness is affected by scaffold properties†
Abstract
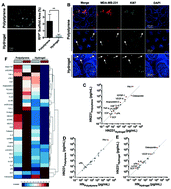
Distant metastasis is the major cause of breast cancer-related mortality, commonly emerging clinically after 5 or more years of seeming ‘cure’ of the primary tumor, indicating a quiescent dormancy. The lack of relevant accessible model systems for metastasis that recreate this latent stage has hindered our understanding of the molecular basis and the development of therapies against these lethal outgrowths. We previously reported on the development of an all-human 3D ex vivo hepatic microphysiological system that reproduces several features of liver physiology and enables spontaneous dormancy in a subpopulation of breast cancer cells. However, we observed that the dormant cells were localized primarily within the 3D tissue, while the proliferative cells were in contact with the polystyrene scaffold. As matrix stiffness is known to drive inflammatory and malignant behaviors, we explored the occurrence of spontaneous tumor dormancy and inflammatory phenotype. The microphysiological system was retrofitted with PEGDa-SynKRGD hydrogel scaffolding, which is softer and differs in the interface with the tissue. The microphysiological system incorporated donor-matched primary human hepatocytes and non-parenchymal cells (NPCs), with MDA-MB-231 breast cancer cells. Hepatic tissue in hydrogel scaffolds secreted lower levels of pro-inflammatory analytes, and was more responsive to inflammatory stimuli. The proportion of tumor cells entering dormancy was markedly increased in the hydrogel-supported tissue compared to polystyrene. Interestingly, an unexpected differential response of dormant cells to varying chemotherapeutic doses was identified, which if reflective of patient pathophysiology, has important implications for patient dosing regimens. These findings highlight the metastatic microphysiological system fitted with hydrogel scaffolds as a critical tool in the assessment and development of therapeutic strategies to target dormant metastatic breast cancer.

 Please wait while we load your content...
Please wait while we load your content...