From recovered metal waste to high-performance palladium catalysts†
Abstract
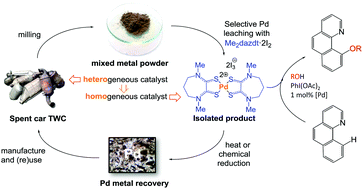
The catalytic activity of a series of neutral and cationic, homo- and heteroleptic, mono- and bimetallic palladium(II) compounds based on dithiocarbamate and dithiooxamide S,S-donor ligands is described. High activity was observed in the regio- and chemo-selective C–H functionalization of benzo[h]quinoline to 10-alkoxybenzo[h]quinoline and 8-methylquinoline to 8-(methoxymethyl)quinoline in the presence of the oxidant PhI(OAc)2. The best performance was found for [Pd(Me2dazdt)2]I6 (Me2dazdt = N,N′-dimethyl-perhydrodiazepine-2,3-dithione), [PdI2(Me2dazdt)] and [Pd(Cy2DTO)2]I8 (Cy2DTO = N,N′-dicyclohexyl-dithiooxamide) which are all obtained directly as products of sustainable Pd-metal leaching processes used to recover palladium from scrap metal. These compounds provided almost quantitative yields under milder conditions (50 °C, 1–3 mol% Pd loading) and much shorter reaction times (1–3 h) than reported previously. These results illustrate how the complexes obtained from the selective and sustainable recovery of Pd from automotive heterogeneous Three Way Catalysts (TWC) can be employed directly in homogeneous catalysis, avoiding further metal recovery steps and valorising the metal complex itself in a ‘circular economy’ model.


 Please wait while we load your content...
Please wait while we load your content...