Life cycle assessment of CO2-based C1-chemicals†
Abstract
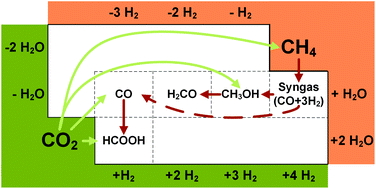
Carbon dioxide (CO2) and hydrogen are promising feedstocks for a sustainable chemical industry. Currently, the conversions of CO2 and hydrogen are most advanced for chemicals with 1 carbon atom, the so-called C1-chemicals, with the first pilot plants in operation. For formic acid, carbon monoxide, methanol, and methane, CO2-based C1-chemicals can reduce the impacts of fossil depletion and global warming through the substitution of fossil-based processes. Existing life cycle assessment (LCA) studies for carbon monoxide, methanol, and methane show that a reduction in environmental impacts is achieved if hydrogen is supplied by water electrolysis with renewable electricity. However, in the foreseeable future, renewable electricity will be limited. Thus, from an environmental point of view, renewable electricity should be employed for chemical processes in the order of highest environmental impact reductions. Environmental impact reductions are the difference in environmental impacts of fossil-based processes and CO2-based processes. In this study, we compared the CO2-based production of formic acid, carbon monoxide, methanol, and methane. We determined the reduction of global warming and fossil depletion impacts using 1 kg of hydrogen. Our results show that the CO2-based production of formic acid achieves the highest environmental impact reductions, followed by carbon monoxide and methanol. The lowest environmental impact reductions are achieved for CO2-based methane production. Our analysis reveals that the CO2-based production of formic acid can reduce environmental impacts, compared to the fossil-based process, even if hydrogen is supplied by fossil-based steam-methane-reforming.


 Please wait while we load your content...
Please wait while we load your content...