Structural diversity in Cu(i) complexes of the PNP ligand: from pincer to binuclear coordination modes and their effects upon the electrochemical and photophysical properties†
Abstract
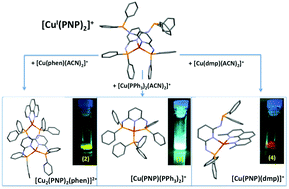
A set of new copper(I) complexes is synthesized and characterized using a labile PNP pincer ligand (PNP = N,N′-bis(diphenylphosphine)-2,6-diaminopyridine). A homoleptic Cu(I) complex [Cu(PNP-κP1:κN1)2]+, (1), was prepared, and taking advantage of the uncoordinated phosphorus atoms in (1), reaction with a second Cu(I) atom bearing secondary ligands (PPh3, phen or dmp) allows the formation of new complexes: a bimetallic helicate [CuI2(PNP)2(phen)]2+, (2), a mononuclear pincer complex [CuI(PNP)(PPh3)]+, (3), and a heteroleptic complex [CuI(PNP)(dmp)]+, (4). All complexes were characterized by X-ray crystallography, NMR (VT-NMR for (1) and (4)), cyclic-voltammetry, and steady-state and time-resolved luminescence spectroscopy. The fluxional behavior in (1) was studied by 31P VT-NMR, where an Ea value of 47.42 kJ mol−1 was calculated for the intramolecular alternating coordination of –PPh2 moieties in PNP to the metal atom. This set of compounds reveals the versatility of the PNP ligand when added to the coordinating properties of Cu(I). The four complexes exhibit emission in solution and complexes (2)–(4) display intense luminescence in the solid state. The oscillographic traces showing the decay of the luminescence were fitted to biexponential functions with time constants: 8.0 μs > τem,1 > 0.37 μs and 50 μs >τem, 2 > 2.2 μs for complexes (2), (3) and (4), respectively. Radiative relaxation is associated with electronic transitions in both the ligand PNP and metal-to-ligand charge transfer (MLCT).


 Please wait while we load your content...
Please wait while we load your content...