Smart photocatalytic removal of ammonia through molecular recognition of zinc ferrite/reduced graphene oxide hybrid catalyst under visible-light irradiation†
Abstract
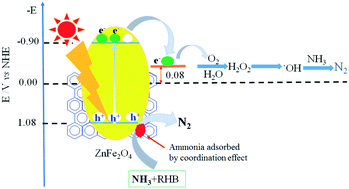
Zinc ferrite loaded with reduced graphene oxide (ZnFe2O4/rGO) and zinc ferrite (ZnFe2O4) catalysts were synthesized via a one-spot method. The catalysts were characterized by X-ray diffraction, transmission electron microscopy, Fourier-transform infrared spectroscopy, Raman spectroscopy, UV-vis diffuse reflectance spectroscopy, surface photovoltage spectroscopy and X-ray photoelectron spectroscopy. Results revealed that the as-synthesized ZnFe2O4/rGO and ZnFe2O4 particles were cubic spinel-type ZnFe2O4 with space group number of Fd3m, and that their average diameters were 7.4 and 7.0 nm, respectively. The photocatalytic results indicated that the ZnFe2O4/rGO hybrid catalyst possesses higher activity than that of the single ZnFe2O4 component under visible-light irradiation. More importantly, the ZnFe2O4/rGO catalyst could recognize ammonia from an organic pollutant-ammonia mixed solution and selectively degrade ammonia and nitrogen gas based on the coordination recognition between Zn cations on ZnFe2O4 and ammonia in solution. Fourier-transform infrared, Raman and X-ray photoelectron spectra confirmed that ammonia was selectively adsorbed on ZnFe2O4 particles. The shifts of Zn 2p3/2 and Zn 2p1/2 binding energies confirmed the coordination between Zn cations and ammonia. The surface photovoltage spectra revealed that the photo-generated holes moved to the surface of ZnFe2O4 particles upon incident visible-light irradiation, and degraded ammonia adsorbed on the catalyst surface. These findings will encourage more investigations of photocatalysis based on coordination recognition.


 Please wait while we load your content...
Please wait while we load your content...