Preparation of zirconium and hafnium complexes containing chiral N atoms from asymmetric tertiary amine ligands, and their catalytic properties for polymerization of rac-lactide†
Abstract
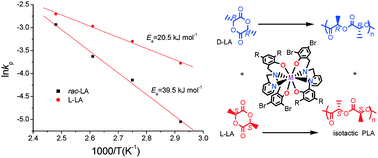
A series of N-(2-pyridylmethyl)-N,N-bis(2′-hydroxy-benzyl) amine ligands were synthesized, and the new asymmetric tertiary amine ligands reacted with ZrCl4 and HfCl4 to give complexes 1–6. The chiral N atoms were created in the complexes upon coordination of the asymmetric tertiary amine ligands to the metal centers. The complexes were used as catalysts for ring-opening polymerization of rac-lactide. The results show that the substituents and structure of the ligands have effects on the activity and stereoselectivity of the catalyst. The stereoselectivity was promoted by the complex containing the ligand with the electron-withdrawing group. Complex 3, with bulkier cumyl groups on the ligand, possesses better stereoselectivity. The complexes are able to catalyze the living control ring-opening polymerization of rac-lactide to afford isotactic-rich polylactide with narrow molecular weight distribution. Kinetic studies reveal that polymerization is of the first order in both monomer and catalyst. The microstructure of the resulting polymer is determined by 1H NMR homonuclear decoupled spectra. The results show that the isotactic-rich chain is dominant in the prepared polylactide. The Pm value is in the range of 0.67–0.81.


 Please wait while we load your content...
Please wait while we load your content...