Formation of C2 oxygenates and ethanol from syngas on an Fe-decorated Cu-based catalyst: insight into the role of Fe as a promoter†
Abstract
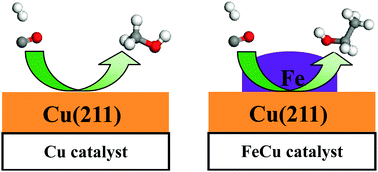
In this study, the formation mechanism of C2 oxygenates and ethanol from syngas on Fe-decorated Cu bimetallic catalyst was investigated using density functional theory (DFT) calculations together with microkinetic modeling. The results showed that CH2 was the most favored monomer among all the CHx (x = 1–3) species over the FeCu bimetallic catalyst, which was more favorable than CH3OH formation. Namely, the FeCu catalyst exhibited a good selectivity toward CH2 formation instead of CH3OH formation in syngas conversion. Starting from the CH2 monomer, CH2CO and CH3CO via CO insertion into CH2 and CH2CO hydrogenation were the major products instead of C2 hydrocarbons or methane, CH3CO was successively hydrogenated to ethanol via CH3CHO and CH3CH2O intermediates. Moreover, the microkinetic modeling showed that the FeCu bimetallic catalyst had a high selectivity toward ethanol rather than methanol and methane. Further, the addition of Fe into the Cu catalyst promoted CHx formation by accelerating C–O bond cleavage, suppressed methanol formation, and facilitated C2 oxygenate formation rather than methane formation, suggesting that the synergetic effect between Fe and Cu played an important role in the formation of C2 oxygenates and ethanol. In addition, it is believed that the insights derived from this study can provide clues for the catalyst design of oxygenate synthesis and other bimetallic catalytic systems.


 Please wait while we load your content...
Please wait while we load your content...