Accurate experimental and theoretical enthalpies of association of TiCl4 with typical Lewis bases used in heterogeneous Ziegler–Natta catalysis†
Abstract
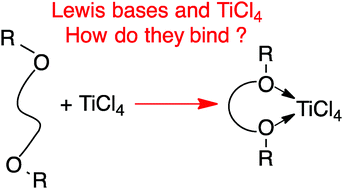
Adducts of TiCl4 with Lewis bases used as internal or external donors in heterogeneous Ziegler–Natta (ZN) catalysis represent a fundamental interaction contributing to the final composition of MgCl2 supported ZN-catalysts. This study presents the accurate experimental evaluation, from titration calorimetry, of the formation enthalpy of TiCl4 adducts with 15 Lewis bases of industrial interest. In addition, we report the accurate energies of association of TiCl4 with the same Lewis bases from calculations at the DLPNO-CCSD(T) level of theory. These accurate experimental and theoretical association values are compared with selected methods based on density functional theory (DFT) in combination with popular continuum solvation models. Calculations suggest that the PBE-D3, and M06 functionals in combination with a triple-ζ plus polarization quality basis set provide the best performance when the basis set superposition error (BSSE) is not removed from the association energies. Cleaning the association energies with the BSSE with the counterpoise protocol suggests B3LYP-D3, TPSS-D3 and M06L as the best performing functionals. The introduction of solvent effects with the PCM and SMD continuum solvation models allows the DFT-based association enthalpies to be compared with the experimental values obtained from titration calorimetry. Both solvation models in combination with the PBE-D3, PBE0-D3, B3LYP-D3, TPSS-D3, M06L, and M06 functionals provide association enthalpies close to the experimental values with MUEs in the range of 10–15 kJ mol−1.


 Please wait while we load your content...
Please wait while we load your content...